Oscilla is a Danish company, which has produced medical equipment for hearing tests since 1959. The Oscilla Audiometer is known around the world for its ease of use and simple design.
Ultra-portable hearing test
The new generation of USB powered Oscilla® audiometers provide an ultra-portable hearing test solution. With a total weight of less than 900 grams and a single plug installation, it is the optimal solution for testing in the field and in satellite clinics.
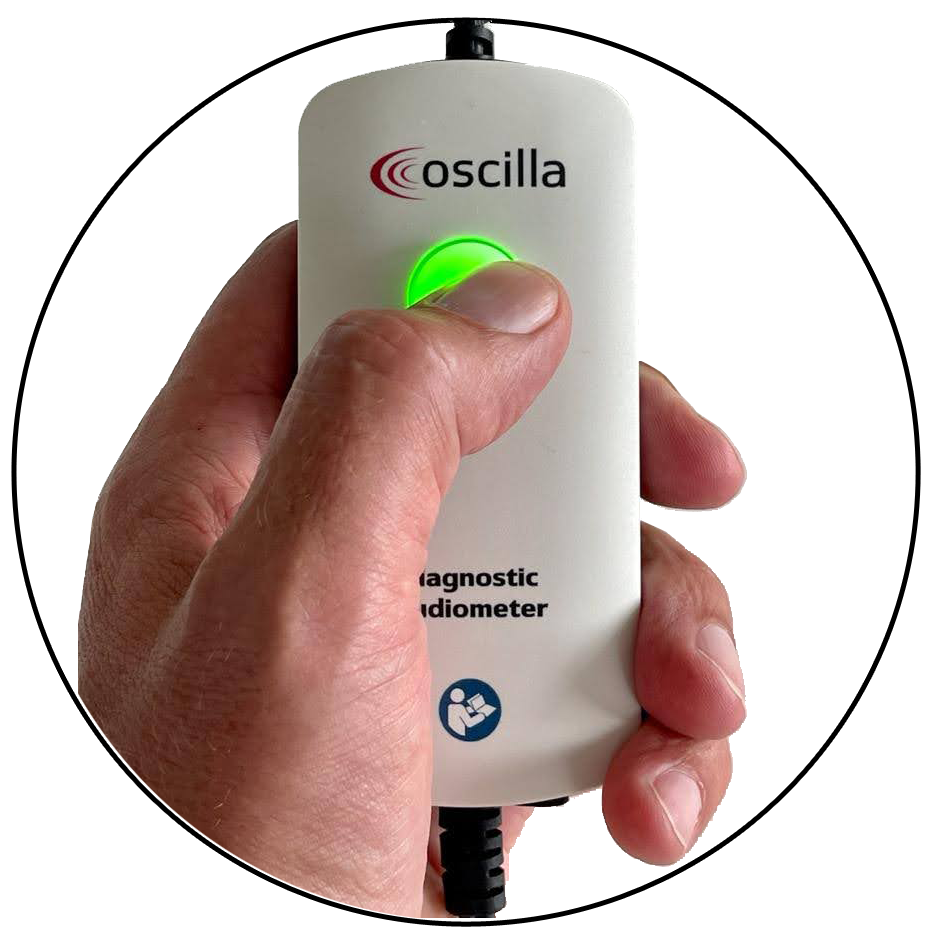
Patient Interaction
The colored luminous button is made of medical grade silicone. It lights up when the devices are connected, and changes color during the session to improve interaction with the patient.
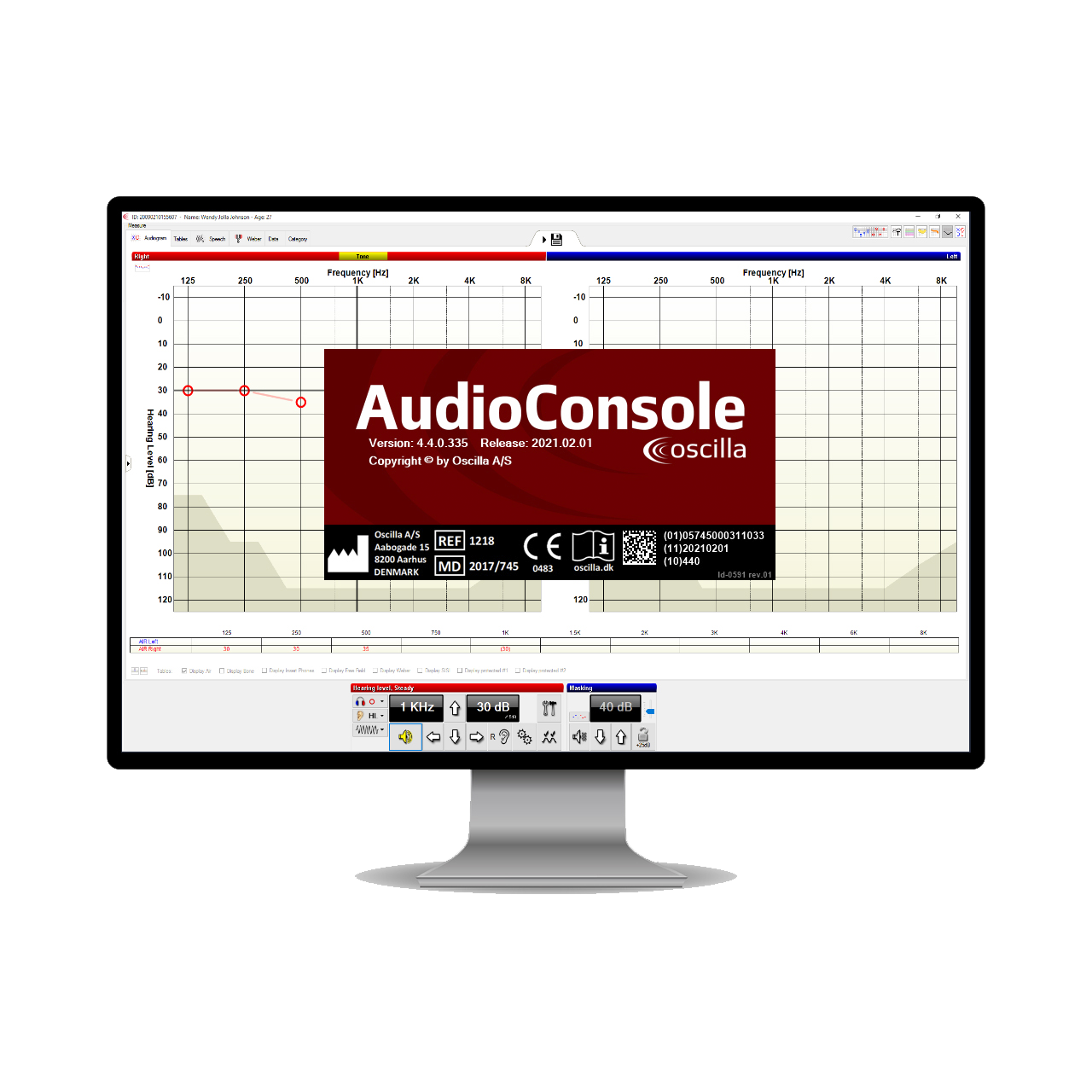
The software
The Oscilla AudioConsole® software is well known for its straightforward workflow and is integrated with many patient management systems around the world. The last version has passed MDR and is now classified as IIa medical device software. it is compatible with new and all existing Oscilla devices.
User Experience Design
In Oscilla, we conduct a User Experience Design process with the sole purpose of designing a system that offers a great user experience. Ease-of-use and a straightforward workflow take high priority in the development of Oscilla products and software.